Back to Dual Nature of Matter and Radiation 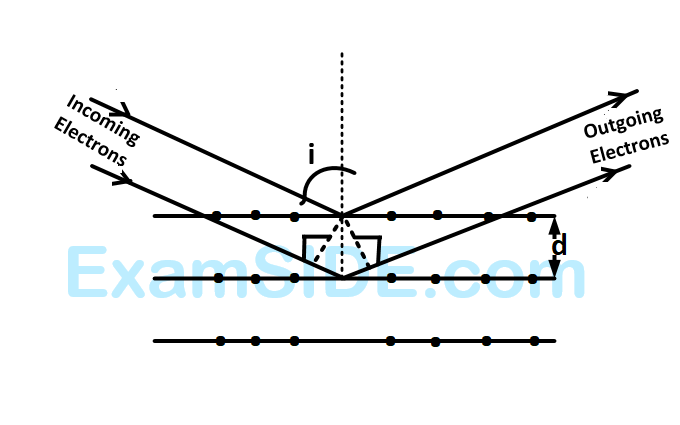
Class 12 • Physics • Chapter-11
Dual Nature of Matter and Radiation
Question 208 of 394
208MediumJEE Mains2008
Wave property of electrons implies that they will show diffraction effects. Davisson and Germer demonstrated this by diffracting electrons from crystals. The law governing the diffraction from a crystal is obtained by requiring that electron waves reflected from the planes of atoms in a crystal interfere constructively (see figure). If a strong diffraction peak is observed when electrons are incident at an angle $'i' from the normal to the crystal planes with distance 'd' between them (see figure), de Broglie wavelength {\lambda _{dB}} of electrons can be calculated by the relationship (n$ is an integer)
